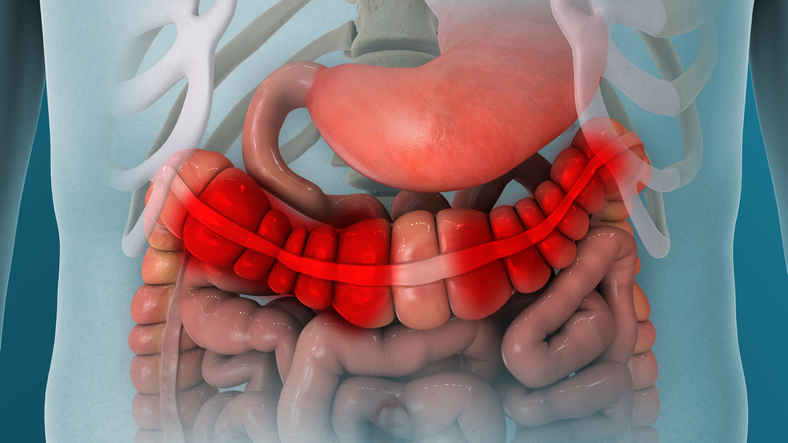
A new drug that was designed using generative artificial intelligence (AI) for the treatment of inflammatory bowel disease (IBD) demonstrated a favorable pharmacokinetic (PK) profile in validating gut-restrictive properties. ISM5411, a hypoxia-inducible factor prolyl hydroxylase (PHD) inhibitor, was found to be generally safe and well tolerated in all dose groups of the phase I trials conducted in Australia and China. Study details and results were published in Nature Biotechnology.
The early discovery, supported by Insilico Medicine’s commercially available generative AI platform Chemistry42, of the preclinical candidate nomination of ISM5411 took only 12 months, during which approximately 115 molecules were synthesized and screened.
“It is pleased to see the good safety and PK profiles of ISM5411, especially the low systemic exposure across all dose groups. These results are suggestive of the gut-restrictive property of ISM5411 and good potential for further clinical development,” said Philip Ryan, MD, PhD, the principal investigator of ISM5411 phase I study in Australia. “As an advanced technology, AI has played important roles in drug discovery and clinical research. We are looking forward to following ISM5411 as it progresses into patient trials, where we hope to see clinical benefits stemming from this technology.”
These studies included single ascending dose (SAD), multiple ascending dose (MAD), and food effects parts involving 76 healthy participants in Australia and 48 healthy participants in China. Safety and PK data collection has been completed for both studies. There were no reports of serious adverse events or treatment-related adverse events leading to discontinuation. ISM5411 also exhibited a favorable PK profile for validating gut-restrictive properties, with very low systemic exposure and a high fecal/plasma ratio in healthy volunteers.
“Thus, we conclude that ISM012-042 is safer than existing PHD inhibitors as it exhibited low systemic inhibition of PHDs,” the researchers noted.
Source





 © 2025 Mashup Media, LLC, a Formedics Property. All Rights Reserved.
© 2025 Mashup Media, LLC, a Formedics Property. All Rights Reserved.