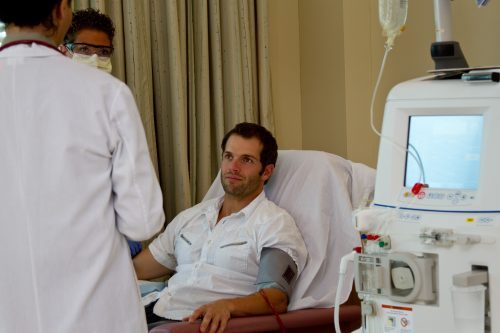
The preferred method for vascular access in patients requiring hemodialysis is arteriovenous fistula (AVF). AVF failure is associated with increased use of catheter access, and the related increase in morbidity and mortality. According to Mohamad Anas Hussain, MD, PhD, and colleagues, there is a need for an alternative option for vascular access that provides the benefits of AVFs, such as low infection rate, while reducing the associated maturation failure possibility.
During an oral session at ASN Kidney Week 2024, the researchers presented results of a phase 3 trial assessing the safety and efficacy of an acellular tissue engineered vessel (ATEV) compared to autologous AVF. (CLN-PRO-V007; ClinicalTrials.gov, NCT03183245). The presentation was titled Prospective Randomized Trial of Humacyte’s Acellular Tissue Engineered Vessel vs Autologous Arteriovenous Fistula for Hemodialysis Access.
The phase 3, prospective, multicenter, two-arm, randomized controlled trial included 242 individuals undergoing surgical vascular access creation for hemodialysis. Participants were randomized to receive either the ATEV (n=123) or autologous AVF (n=119). Primary outcomes of interest included 6-month functional patency and 12-month secondary patency following access creation. Secondary outcomes were comparisons of access usability and infection rates.
The mean age of the overall cohort was 58.6 years and 29% were female. Participants in the ATEV group had higher rates of functional patency at both six months (81% vs 68% for AVF) and 12 months (68% vs 62% for AVF), P=.0071; joint test for the coprimary endpoints. The mean duration of ATEV use was significantly higher than duration of AVF use (7.5 months vs 6.1 months, respectively; P=.0162).
In the subgroup of female participants, the rates of 6-month functional patency were higher among ATEV users compared to those with AVF access (89% vs 55%, respectively), as were 12-month functional patency rates (81% vs 49%, respectively); P<.001; joint test).
Infection rates related to the access type were comparable (5.8% ATEV vs 4.1% AVF). No unexpected safety events were observed.
“Humacyte ATEV had better access functional patency and usability versus AVFs at one year,” the authors said. “Female subjects experienced improved outcomes, supporting ATEV consideration as a novel access option for subjects at high risk for AVF non-maturation.”
Source: Hussain MA, Ozaki CK, Moore EE, Khondker Z, Parikh SJ, Niklason, LE. Prospective randomized trial of Humacyte’s acellular tissue engineered vessel vs autologous arteriovenous fistula for hemodialysis access. SA-OR95. Abstract of an oral presentation at the American Society of Nephrology Kidney Week 2024; October 26, 2024; San Diego, California. Funding was provided by Humacyte Global, Inc.



 © 2025 Mashup Media, LLC, a Formedics Property. All Rights Reserved.
© 2025 Mashup Media, LLC, a Formedics Property. All Rights Reserved.