
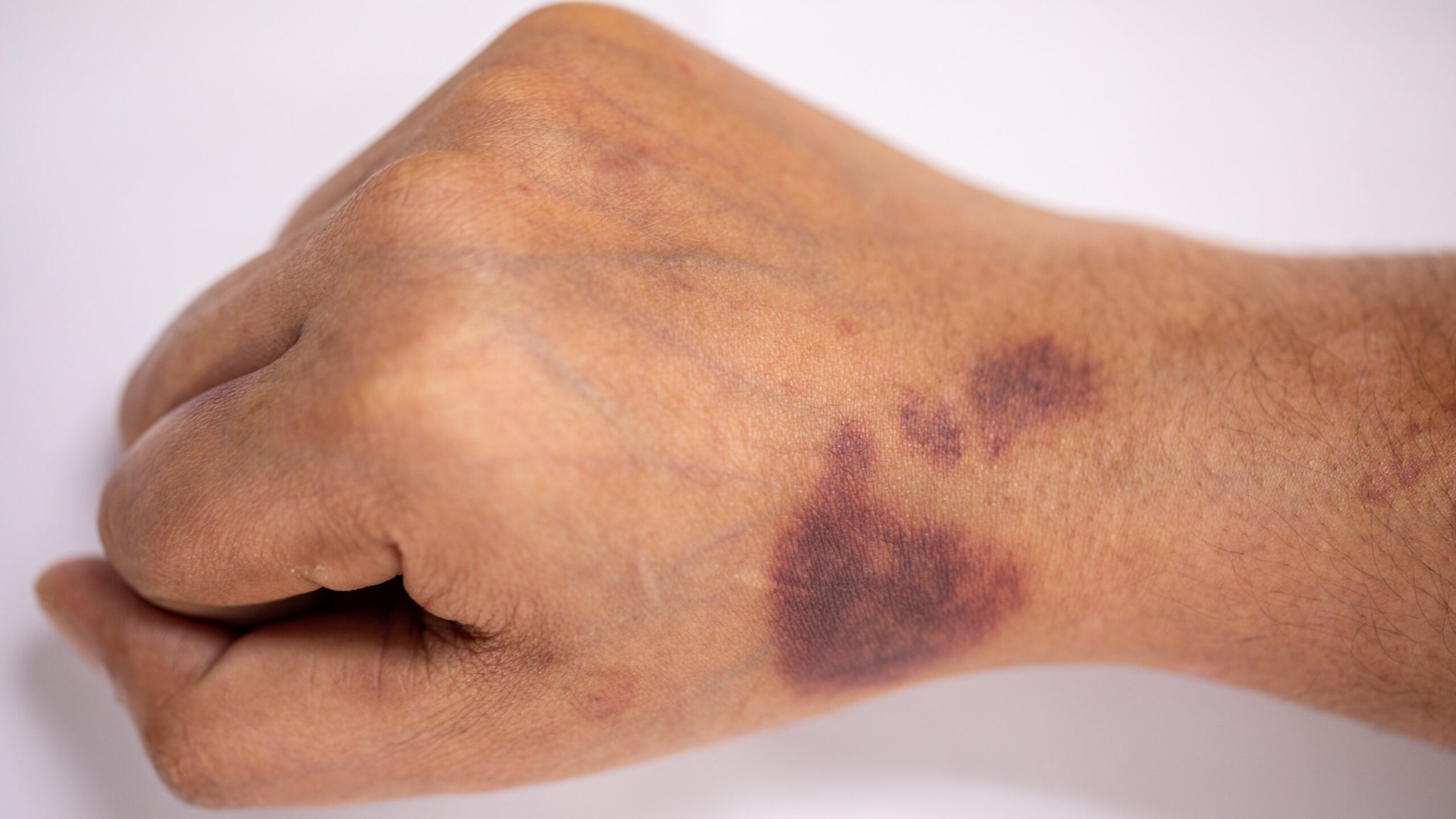
Hemophilia
Advertisement
ASH warns NIH cuts would jeopardize blood disorder care and halt key progress in hematology research.
A preclinical mouse study showed hemostasis restoration, as well as use for active bleeding and internal bleeding prevention.
Fidanacogene elaparvovec gene therapy is a safe and effective treatment for patients with hemophilia B, research shows.
Approval was based on data from phase 3 studies in the ongoing ATLAS clinical development program.
The Clinical Practice Guidance document issued by the EHA, ISTH, EAHAD, and ESO addresses both hemophilia A and B.
A study cohort with severe disease over a median follow-up of 14 months had an annualized bleeding rate of zero.
This approval for patients aged 12 years and older was based on findings from the explorer7 phase III clinical trial.
A new study addresses the need for data on treatment outcomes in patients younger than 12 years of age.
Quality of life improvement is among the benefits from this agent in patients with congenital disease.
A retrospective observational study's clearest takeaway was that the agent can reduce spontaneous bleeding in severe disease.
These proof-of-concept data may spur further studies toward developing a single-intervention cure for patients.
Patients with hemophilia A who received prophylaxis had a reduced risk for ICH.
Analysis shows pharmacokinetic-guided dosing may be effective for guiding prophylactic factor concentrate therapy.
Compared with before surgery, the percentage of factor VIII increase was 42.9% at day one and 36.4% on day two.
Free tissue factor pathway inhibitor levels were associated with thrombin generation in patients with severe hemophilia.
The US FDA has approved fidanacogene elaparvovec-dzkt, a gene therapy for adults with hemophilia B.
Dr. Mamauag details her study on the need for factor VIII infusions in patients with tolerized hemophilia A on emicizumab.
The US FDA has approved an expanded indication for IXINITY to control bleeding in all patients with hemophilia B.
Desmopressin with FVIII during operations was reportedly feasible and safe in patients with nonsevere hemophilia A.
Patients with hemophilia A or B had low rates of VTE after total joint replacement despite infrequent thromboprophylaxis use.
Advertisement









 © 2025 Mashup Media, LLC, a Formedics Property. All Rights Reserved.
© 2025 Mashup Media, LLC, a Formedics Property. All Rights Reserved.